The past century has witnessed the unprecedented industrial production and wide application of plastics. Their versatility and low cost have made themselves a class of indispensable materials in the modern society. However, the “white pollution” issues are meanwhile increasingly apparent with the huge consumption of single-use packaging materials in daily life which account for almost 40% of plastics produced currently. To make matters worse, the vast majority of commercial plastics are non-degradable, which would cause long-term and lasting pollution to the environment. Precisely for this, one of the feasible solutions is to develop alternative degradable plastics, as proposed by polymer chemists.
Based on the final degraded products, two main degradable strategies are involved, namely disposing and decomposing to small molecule (DDM) and chemical recycling to monomer (CRM). Specifically, polymers undergoing DDM are featured with labile backbones and can degrade into non-toxic small molecules by external stimuli such as pH, temperature, enzymes and oxidizing or reducing agents. Polyacetals, a type of pH-responsive polymers, are degradable under acidic conditions, while highly stable under neutral and basic circumstances. Polyacetals can thus be degraded by DDM. On the other hand, CRM is a novel strategy to recycle plastics, by which polymers can depolymerize back to monomers under certain conditions and the recycled monomers can polymerize again. As for their synthesis, the cationic ring-opening polymerization (CROP) of cyclic acetals is an elegant and promising approach, suffering fatal side reactions and polymerization-depolymerization equilibrium. This poses a great challenge to the synthesis of high molecular weight polyacetals. That is why this method has been rarely reported in the past two decades. Recent development in CRM restimulates our interests in the long-forgotten CROP method due to its inherent depolymerization characteristics, because the cationic ring-opening polymerization (CROP) of cyclic acetals, one of the major methods for polyacetal synthesis, is an equilibrium reaction with inherent reversibility, which serves as a natural CRM procedure for closed-loop recycling. In summary, polyacetals are recyclable materials with both DDM and CRM potentials.
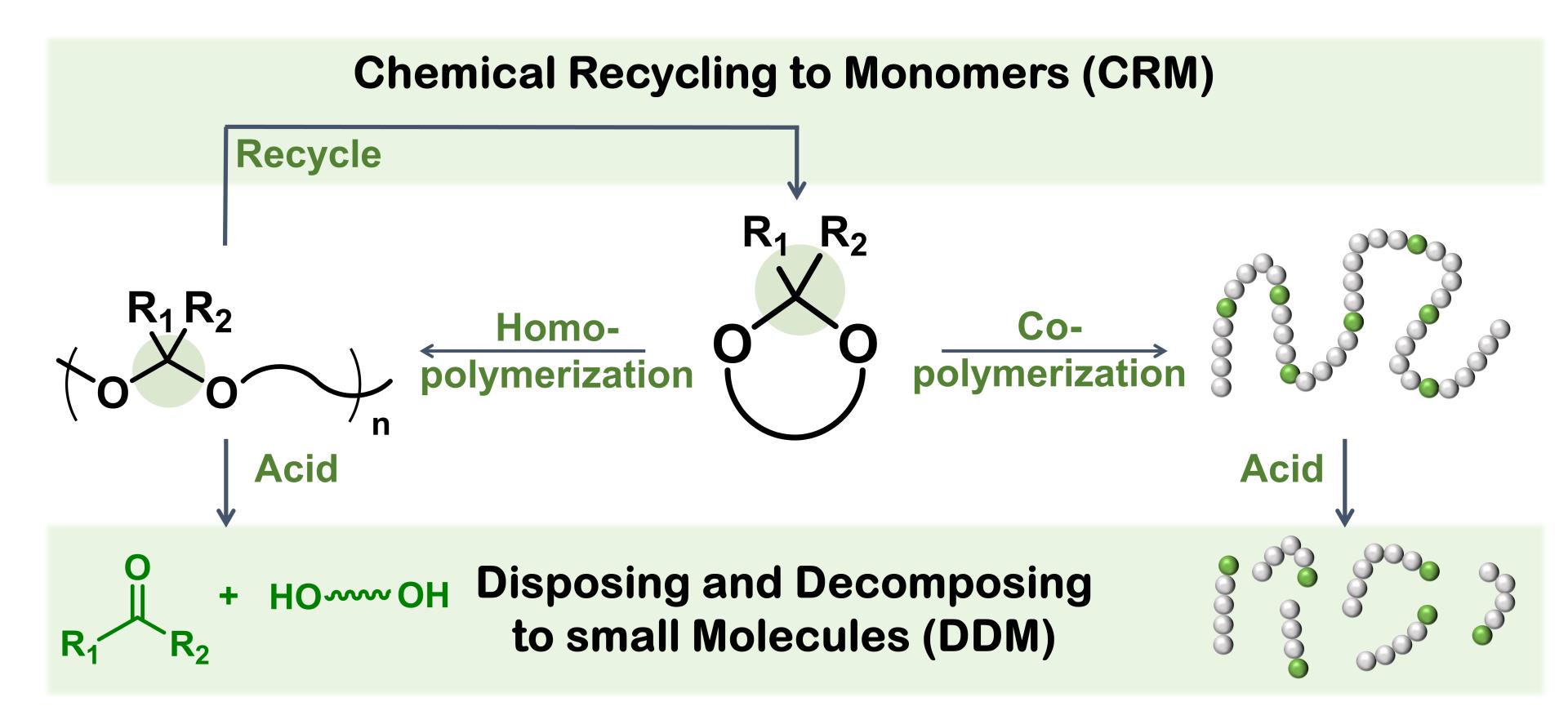
Figure 1. CRM and DDM degradation and recycling strategies of polyacetals
In this review, the authors try to provide a perspicuous and systematic overview on the CROP-made polyacetals from the aspects including polymerization and depolymerization of cyclic acetals, copolymerization of cyclic acetals with heterocyclic or vinyl monomers, degradation and recycling properties of the resulting polymers, with the aim to highlight the DDM and CRM potential of polyacetals as well as to address the key factors for controllable CROP of cyclic acetals (Scheme 1).
To realize the recycling of polyacetals, the premise is to solve the synthetic issues of high molecular weight polyacetals. In this review, the authors discussed the key factors for the synthesis of high-molecular-weight polyacetals by CROP of cyclic acetals from the aspects of initiators, terminators, and polymerization conditions. In addition to CROP of cyclic acetals, this review also introduces the preparation of polyacetals by ring-opening metathesis polymerization of acetal-containing cyclic olefins and cationic/anionic ring-opening polymerization of hemiacetal esters. Recent studies reveal that most of the most commonly used degradable aliphatic polyesters, such as poly(-caprolactone) (PCL), poly(lactic acid) (PLA) and poly(3-hydroxybutyrate) (P3HB), actually do not show obvious degradation even after 1 or 2 years under ambient environment such as in soil and seawater. For the reason, a measure to install acetal groups into such slowly degradable polyesters and non-degradable polyolefins can greatly upgrade the degradation rate and thus promote the DDM process.
The authors thus reviewed the copolymerization of cyclic acetals with heterocyclic compounds (lactones, lactides, cyclic acetals, phosphites, etc.), and vinyl monomers (vinyl ethers, vinyl silyl ethers, vinyl acetate, etc.). Copolymerization of cyclic acetals with cyclic esters and vinyl monomers is a feasible method to improve the degradability of classic polyesters and polyolefins by tuning the chemical structure of the copolymers and their degradation properties.
The review paper is published on Macromolecular Rapid Communications, in which Shen Ting, a doctoral student majoring in polymer chemistry and physics at the Institute of Polymer Science, Zhejiang University, is the first author. Assistant Professor Yougen Chen of Shenzhen University and Professor Jun Ling of Zhejiang University are the corresponding authors.
This work was supported by the National Natural Science Foundation of China and Science and Technology Innovation Commission of Shenzhen.
Paper link: https:// https://onlinelibrary.wiley.com/doi/full/10.1002/marc.202300099


