In March 2026, the research team led by Researcher Lei Qin from the Institute for Advanced Study of Shenzhen University, in collaboration with Professor Yiying Wu’s team from the Ohio State University, published a research paper entitled "Realizing anode-free potassium-organic batteries via sacrificial potassium superoxide additives" in the journal Energy Storage Materials. For the first time, this work introduced potassium superoxide (KO2) as a sacrificial cathode additive into the anode-free potassium-organic battery system, innovatively addressing the core challenge of the initial potassium ion source scarcity in such batteries. Meanwhile, a low-cost and high-safety chemical synthesis route for KO2 was developed, which greatly lowered the threshold for the practical application of anode-free potassium batteries. The Institute for Advanced Study of Shenzhen University is the first completing unit. Yishuo Li is the first author of the paper, and Researcher Lei Qin and Professor Yiying Wu serve as the co-corresponding authors.
Anode-free potassium-organic batteries combine the advantages of organic cathodes, such as environmental friendliness and low cost, with the features of anode-free architectures including high energy density and high safety, thus showing enormous application potential in the field of large-scale energy storage. However, neither the cathode nor the anode of this system pre-stores potassium ion sources, and traditional cathode compensation additives suffer from slow decomposition kinetics, low compensation capacity, and harmful gas release, which have become the key bottlenecks restricting their development. As the sole discharge product of potassium-oxygen batteries, KO2 possesses thermodynamic and kinetic stability. Its decomposition only releases potassium ions and oxygen without any harmful solid residues, making it an ideal candidate material for potassium ion compensation.
In this study, KO2 was preloaded on the organic cathode (PTCDA) as a sacrificial potassium ion source for the first time. Relying on the migration and transfer of soluble superoxide anions, its unique solution-mediated decomposition mechanism achieved a theoretical capacity utilization rate of 97.9 % without additional conductive agents, with a decomposition overpotential below 190 mV, demonstrating excellent reaction kinetics. Differential electrochemical mass spectrometry (DEMS) confirmed that the decomposition of KO2 is a single-electron transfer process with an extremely low carbon dioxide release, which ensures the electrochemical stability of electrodes and electrolytes. At the same time, the team developed a mild new chemical synthesis method for KO2, which can prepare high-purity KO2 in only 5 minutes at room temperature with a cost merely one-third of that of commercial KO2, integrating high efficiency and economy.
The anode-free Cu||PTCDA@KO2 battery constructed based on this strategy successfully realized reversible cycling by depositing potassium ions released from the decomposition of KO2 on the bare copper current collector to form a potassium anode. It still retained a specific capacity of 84.9 mAh/g after 300 cycles, with an average coulombic efficiency as high as 99.5 %. This system is highly compatible with the existing dry-room manufacturing process.
This study for the first time realized the application of KO2-based sacrificial additives in anode-free potassium organic batteries. It not only provides a brand-new idea for solving the problem of initial ion source scarcity in anode-free alkali metal-ion batteries but also promotes the practical development of superoxide-based cathode compensators, laying an important theoretical and experimental foundation for the design and development of next-generation high-energy-density and low-cost energy storage batteries.
This research was supported by the Shenzhen Science and Technology Program (JCYJ20250604182101002).
Link to the original paper: https://doi.org/10.1016/j.ensm.2026.105019
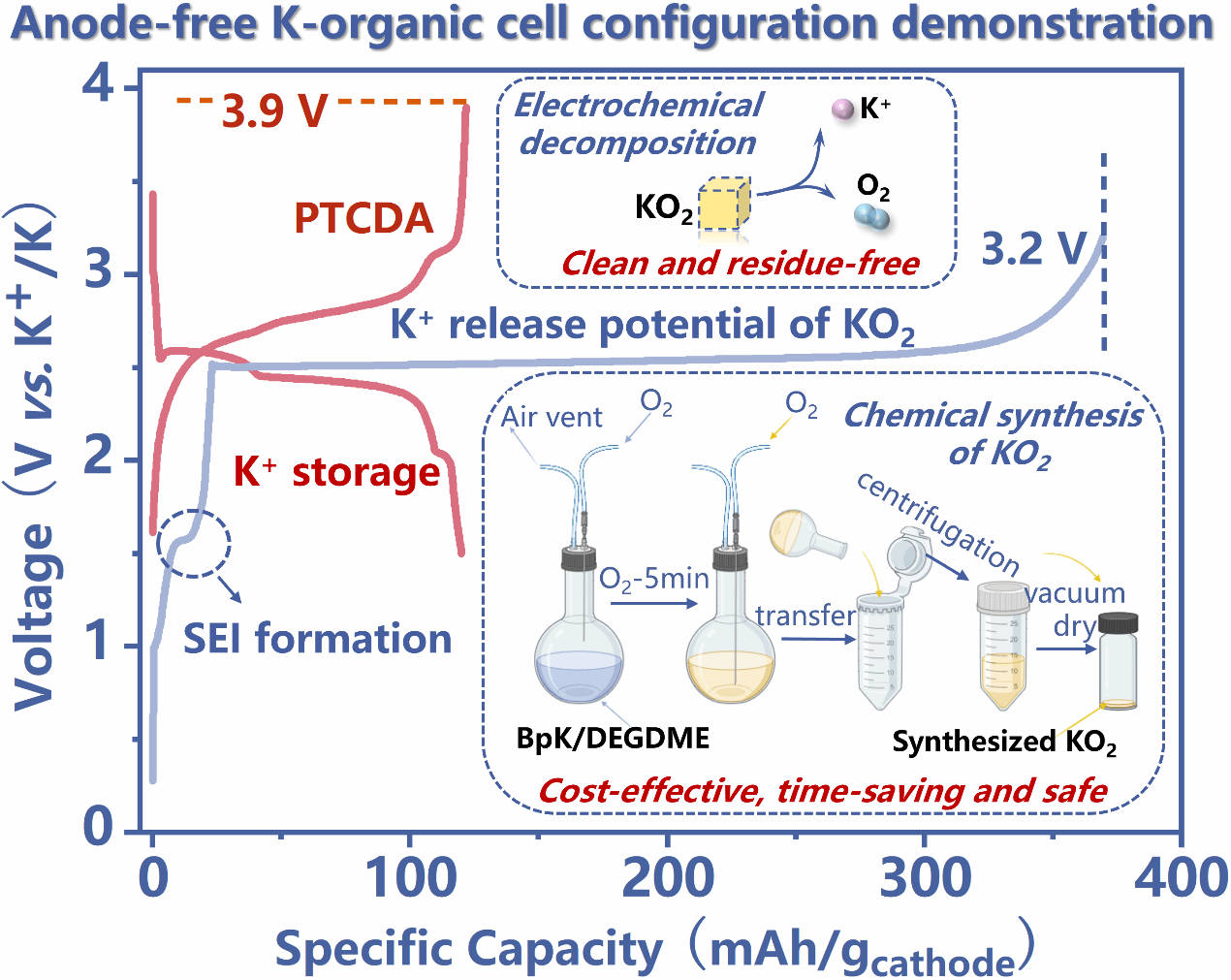
Figure 1 Graphic Abstract.


